A mystery about hormone regulation in pancreatic islet cells is solved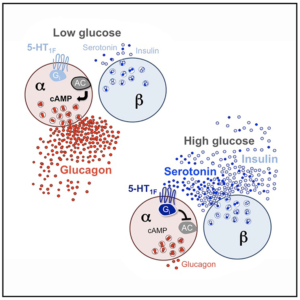 with cADDis!
with cADDis!
Researchers at University of Miami made good use of Montana Molecular’s cADDis assay for cAMP to study how islet cells respond to glucose. This elegant article in Cell Reports authored by Joana Almaça and her colleagues at the University of Miami describes how cAMP signals were captured in live human pancreatic islet cells using cADDis, one of Montana Molecular’s suite of fluorescent biosensor assays for cell signaling. A key feature of fluorescent biosensors is that they can be easily deployed in live primary cultures of cells, such as human pancreatic islets, providing insight into the hidden mechanisms of disease.
With the ability to investigate signaling in the human islet, the Miami research team determined that serotonin is synthesized and released from beta cells under normal physiologic conditions. By manipulating serotonin production in the islet, they were able to unravel the effects of serotonin on hormone secretion and cAMP signaling to demonstrate that serotonin is a paracrine regulator of alpha cell activity.
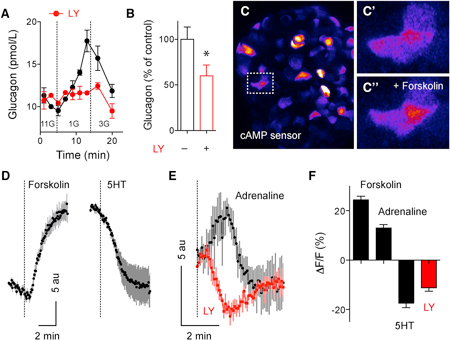
The bright fluorescence produced by cADDis, documented in Figure 5 of the publication (shown below), was key to showing that serotonin released by beta cells lowers cAMP in the neighboring alpha cells. Because cADDis indicates dynamic changes in cAMP, the Miami researchers were able to see that serotonin produces a unique effect. Unlike other paracrine regulators, serotonin produces a rapid and direct decrease in cAMP, quickly shutting down glucagon secretion in response to rising glucose.
According to the authors, the insight gained by measuring cAMP in live islets points to a new approach to treating diabetes with drugs that modulate serotonin signaling in alpha cells.
Many thanks to Vlad Slepak for notifying us about this publication! We sincerely love reading about how our products help researchers understand disease mechanisms leading to new therapeutic opportunities.
Interested in reading about diacylglycerol signaling in islets? See an eariler paper by Patric Rorsmann’s group at OCDEM (Oxford Center for Diabetes, Endocrinology and Metabolism): GLP-1 stimulates insulin secretion by PKC-dependent TRPM4 and TRPM5 activation. (J Clin Invest. 2015)