Optogenetics
Optically elevate intracellular cAMP, measure changes in cAMP and calcium, and create excitable HEK cells.
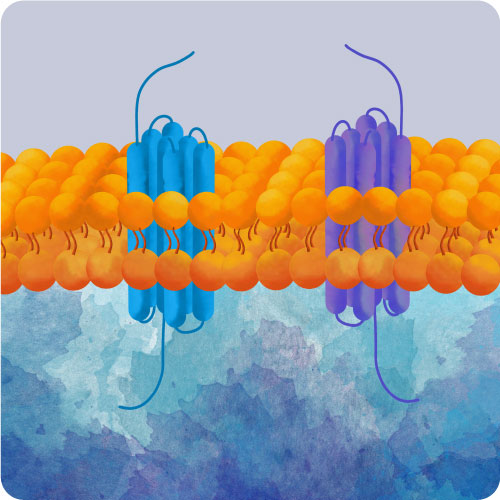
Optogenetics is a rapidly emerging field in which biologists move beyond simply measuring and recording changes in cell signaling (Zhang and Cohen 2017). Now there are tools that enable you to use light to control cellular signaling as well as watch it. Montana Molecular has a growing optogenetics toolkit, all packaged in BacMam virus for low cell-to-cell variability, a simple workflow, and efficient expression in most mammalian cells, including primary and iPSC cultures.
Control & Measure cAMP with Light
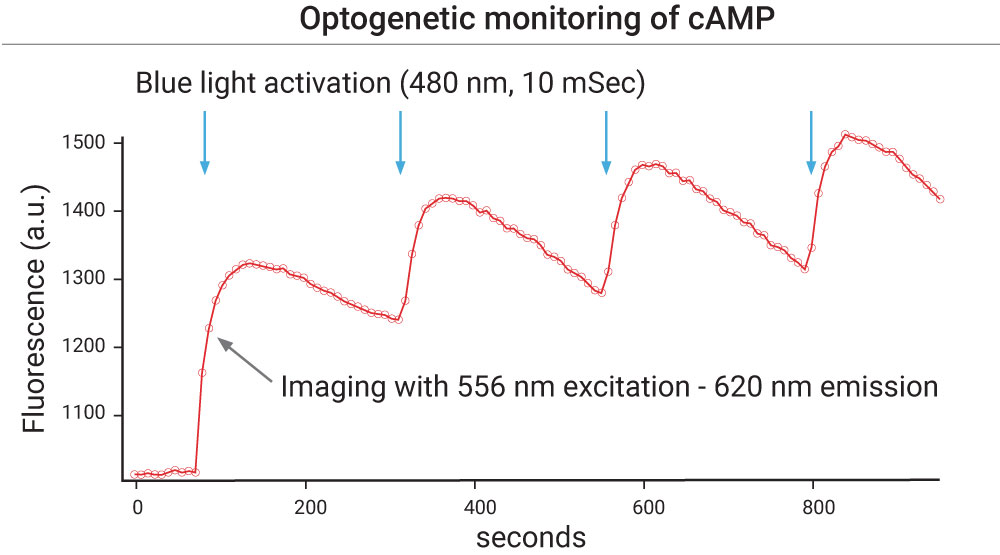
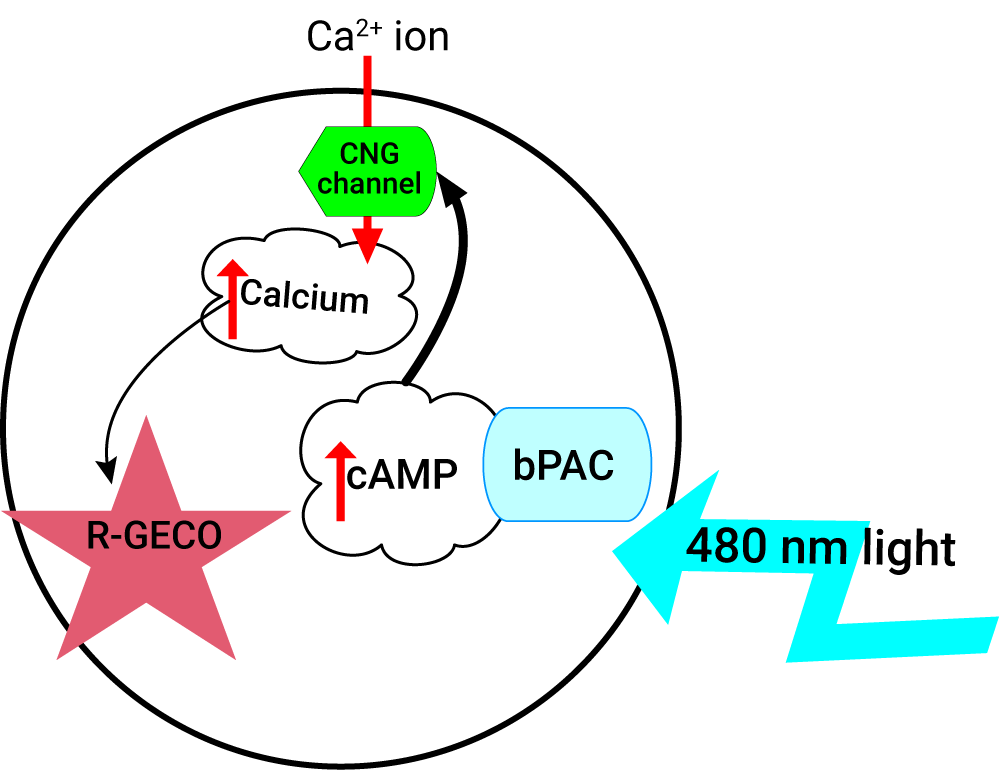
The light activated bPAC enzyme (#V0100N) is an adenylyl cyclase that, when stimulated with faint blue light will rapidly create cAMP. This remarkable protein is incredibly sensitive, and, when not exposed to blue light, shuts down quickly. The action spectra of this protein is in the blue, and light with wavelengths longer than 540 nm do not activate it. This means that the red cADDis (#U0200R) can be co-expressed in the same cell to watch the increases in cAMP in real time. 480 nm light can be used to activate the enzyme, while 560 nm excitation and 590-650 emission can be used to record the changes in fluorescence.
Control cAMP & Measure Ca2+ with Light
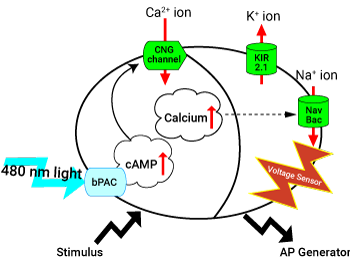
Optically Conrollable, Excitable HEK293 Cells
The addition of a few channels can convert the quiescent HEK 293 cell line into an excitable platform to test voltage sensitive dyes or probes. First, the addition of the KIR 2.11 channel (#V0101N) hyperpolarizes the cell. Second, the voltage sensitive sodium channel (#V0102N) can generate depolarizations. Finally, the light activated bPAC (#V0100N) can be paired with the cAMP-gated channel (#V0103N) such that brief exposures of blue light are sufficient to create spontaneous activity.
Click the link to read this Featured Publication:
Optogenetic Toolkit
Shop Assay Kits by Category
Contact Us
If you have any questions about using these tools in your cells or would like to request a quote, send us an email at info@montanamolecular.com. We’re happy to help!