Live Cell Calcium Assay Kit – GECO
Real time kinetic Ca2+ sensor for fluorescent plate readers and imaging systems.
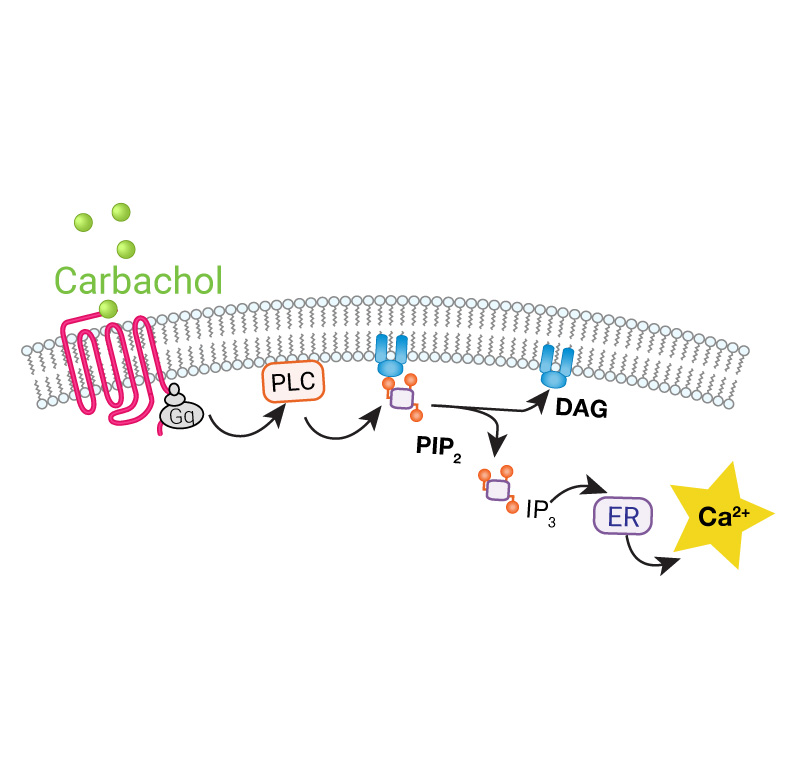
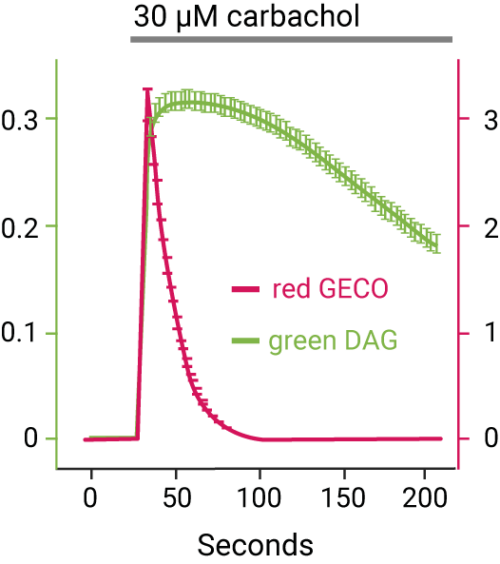
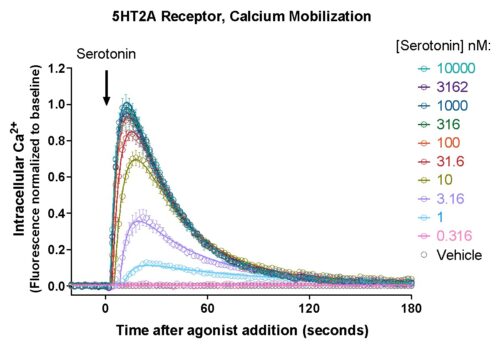
The GECO assays (R-GECO, G-GECO) are fluorescent calcium assays for real time kinetic readouts. This genetically encoded fluorescent sensor is available in both red or green versions, can be expressed in a variety of cell types for plate reader or imaging applications. Developed by Robert Campbell’s lab at University of Alberta, Montana Molecular packages these calcium sensors in viral vectors for robust expression in your favorite cells.
GECO Assay Performance
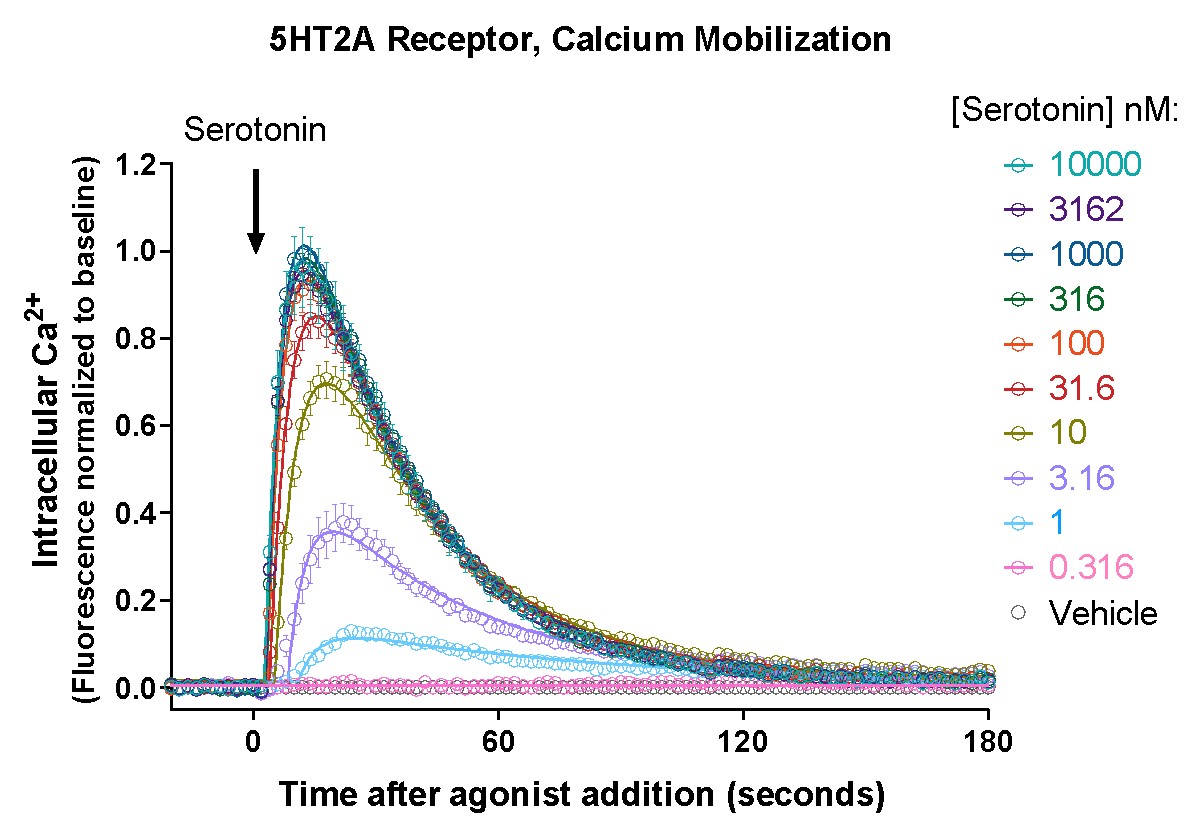
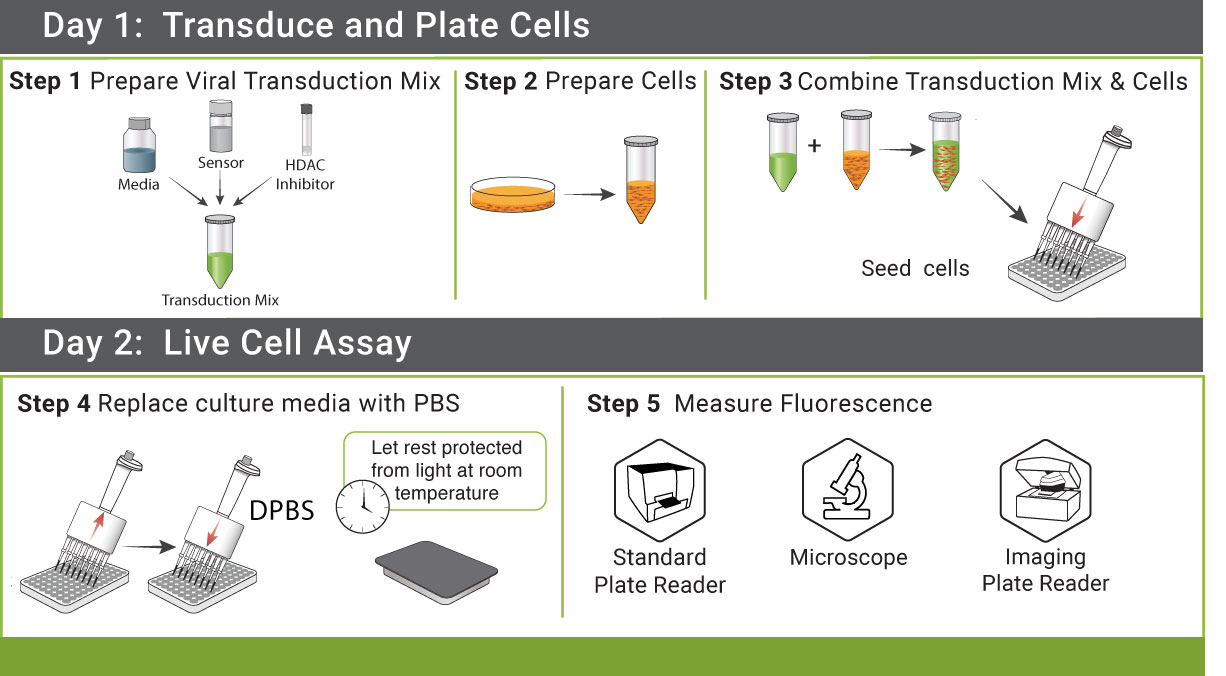
Off the shelf kits include the GECO sensor in BacMam, a BSL-1 viral vector for efficientl delivery to most cell types. Purified BacMam, AAV, Lenti vectors by request.
- Single-channel readout is detectable on standard fluorescence plate readers or imaging systems.
- Robust expression in wide variety of cell types
- Combine with Gs/Gq assay for calcium signaling specificity
- Can be targeted to mitochondria or other microdomains
Bright Red & Green Fluorescent Calcium Sensors
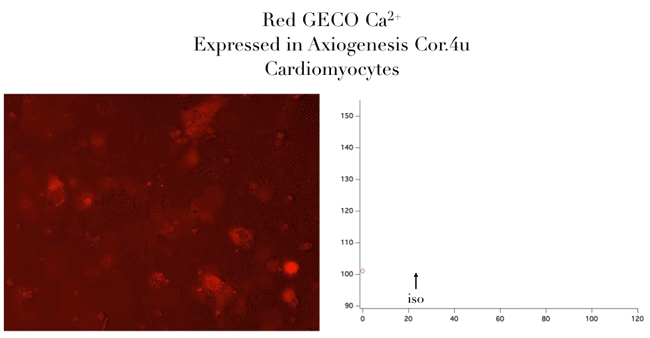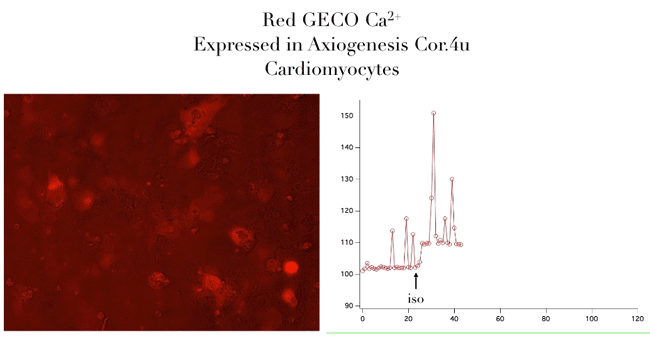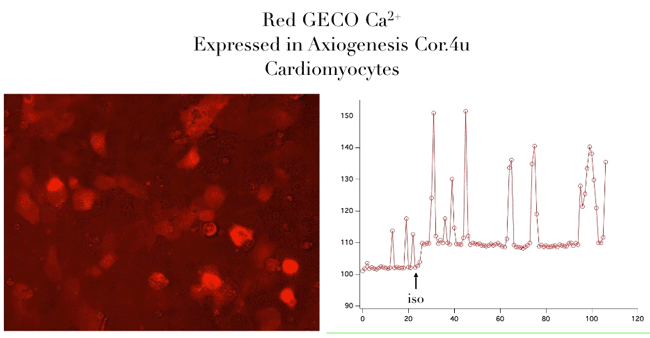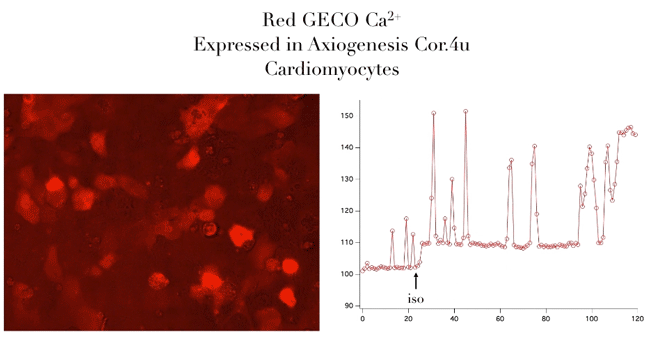
- Genetically encoded for low toxicity
- Real-time kinetic readout
- Robust expression in variety of cell types
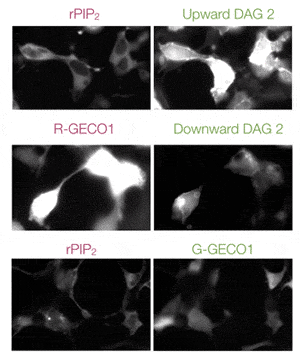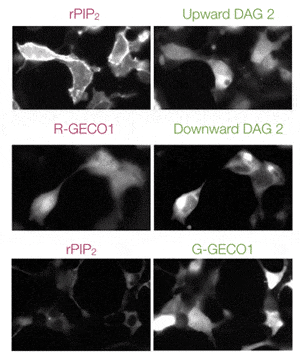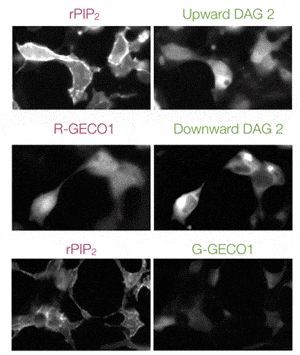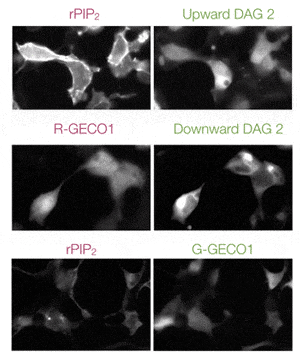
Multiplex Ca2+Assays
Easily combine red or green GECO sensors with other GPCR sensors to measure multiple pathways simultaneously
Testimonials
“Assay is working beautifully with cardiomyocytes, showing a strong and immediate response.”
“Expression in human islets is pretty superb, after 18 hours of incubation. An excellent probe!”
Recent Publications
- X. Chen, et al. Roles for PKC signaling in chromaffin cell exocytosis. Biophysical Journal. December 2024.
- X. Chen, et al. In vitro and in vivo inhibition of the host TRPC4 channel attenuates Zika virus infection. EMBO Molecular Medicine. July 2024.
- X. Chen, et al. A PACAP-activated network for secretion requires coordination of Ca2+ influx and Ca2+ mobilization. Molecular Biology of the Cell. May 2024. (bioRxiv)
- C. Amos, et al. Membrane lipids couple synaptotagmin to SNARE-mediated granule fusion in insulin-secreting cells. Molecular Biology of the Cell. December 2023.
- J. Wu, et al. Interaction Between HCN and Slack Channels Regulates mPFC Pyramidal Cell Excitability and Working Memory. bioRxiv. March 2023.
- M. Thomas, et al. Optically activated, customizable, excitable cells. PLOS One. December 2020.
- L. Liu, et al. Diacylglycerol kinases regulate TRPV1 channel activity. Journal of Biological Chemistry. April 2020.
- S. Hoare, et al. A kinetic method for measuring agonist efficacy and ligand bias using high resolution biosensors and a kinetic data analysis framework. Nature Scientific Reports Feb 2020.
- K. Harlen, et al. Live-Cell Assays for Cell Stress Responses Reveal New Patterns of Cell Signaling Caused by Mutations in Rhodopsin, α-Synuclein and TDP-43 Front. Cell. Neurosci.,December 2019
Posters
TRPML1-Lysosomal-Ca2+ sensor (TLC) for the Ca2+ Dyshomeostasis Neurodegeneration Model
Here we present a genetically encoded TRPML1 Lysosomal Ca2+ (TLC) sensor that enables direct measurement of Ca2+ release with high specificity. Using this tool we show that small molecule agonists differ in their ability to selectively activate TRPML1, with MSK-83 demonstrating higher specificity compared to ML-SA5. The Assay provides a powerful approach for interrogating TRPML1 function in human cells and for identifying modulators of lysosomal Ca2+ signaling.
Perturbed Ca2+ signaling leads to synaptic loss, amyloid and tau aggregation, mitochondrial dysfunction, lysosomal alkalinization, and superoxide production, all of which are hallmarks of neurodegenerative diseases. Evidence from Alzheimers models suggest that ER-derived Ca2+ signals disrupt mitochondria-lysosome interactions, further exacerbating cellular stress.
Measuring the kinetics of G-protein-coupled receptor signaling and biased agonism
Biosensors for G-protein signaling & arrestin recruitment
- Genetically-encoded fluorescent biosensors enable continuous recording of signaling kinetics in live cells
- Gs, Gi, Gq and arrestin
- Easy workflow – add ligand and read in plate reader
A streamlined industrial biosensor platform for quantifying signaling dynamics for drug discovery
Signaling dynamics is a new frontier in GPCR drug discovery, involving the design of drug molecules that signal for the right time in the right place. Montana Molecular has developed biosensors of GPCR signaling with unprecedented performance, enabling sub-second read frequency, total read times of over five hours, and miniaturization to 384-well format, for Gs, Gi, Gq and arrestin pathways.
GPCR Biology
Increase your understanding of drug effects and GPCR biology with bright fluorescent assays for Gs, Gi, and Gq signaling in living cells.