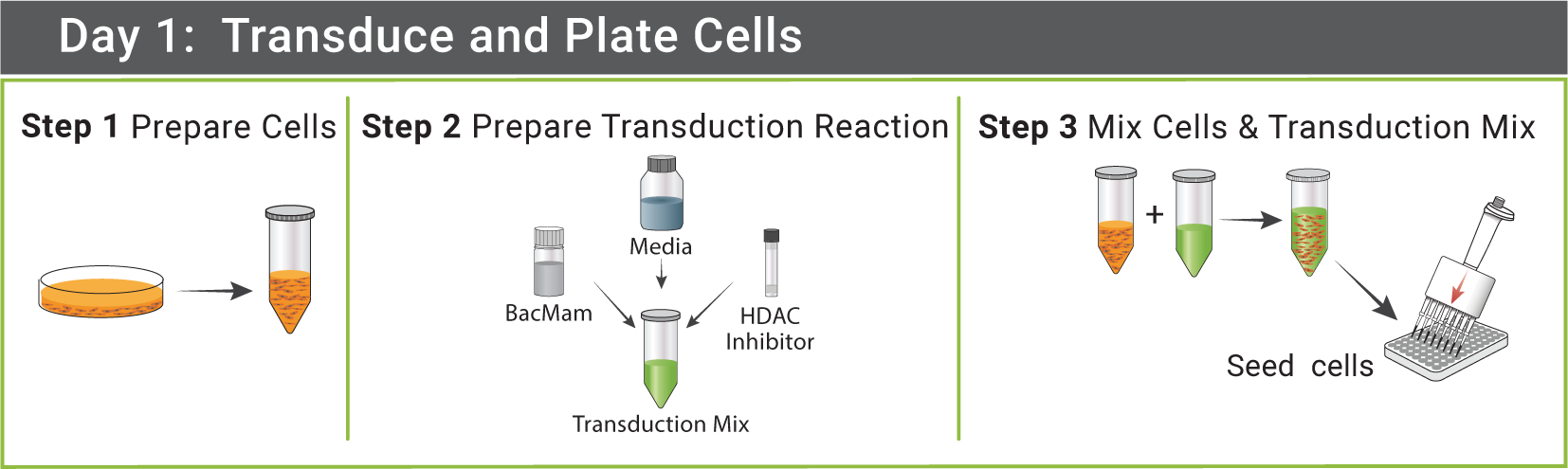
GPCRs in BacMam
Express GPCRs in Mammalian Cells
- Unmodified, untagged, human GPCRs
- Packaged in BacMam for consistent, titratable expression in most mammalian cell types – including cell lines, iPSCs, and primary cultures
- Easily co-express with biosensors for GPCR signaling, GRKs, and RAMPs
- Custom viruses, including splice or species variants, cell-specific promoters, genetic tags, and more are available on request
GPCR Catalog
Ready-to-ship kits
Full List
#Z0100N: Human D1 Dopamine Receptor (DRD1)
#Z0119N: Human D2 Dopamine Receptor (DRD2)
#Z0129N: Human D3 Dopamine Receptor (DRD3)
#Z0209N: Human M1 Muscarinic Receptor (CHRM1)
#Z0200G: Human M1 Muscarinic Receptor (CHRM1) – Green
#Z0200R: Human M1 Muscarinic Receptor (CHRM1) – Red
#Z0219N: Human M2 Muscarinic Receptor (CHRM2)
#Z0220N: Human M3 Muscarinic Receptor (CHRM3)
#Z0309N: Human A1 Adenosine Receptor (ADORA1)
#Z0400N: Human Succinate Receptor (SUCNR1)
#Z0500N: Human β2 Adrenergic Receptor (ADRB2)
#Z0500G: Human β2 Adrenergic Receptor (ADRB2) – Green
#Z0500R: Human β2 Adrenergic Receptor (ADRB2) – Red
#Z0609N: Human GLP-1 Receptor (GLP1R)
#Z0610N: Human Calcitonin Receptor (CALCR)
#Z0620N: Human Vasopressin Receptor (AVPR2)
#Z0709N:Human Nociceptin Opioid Receptor (OPRX)
#Z0719N: Human Kappa Opioid Receptor (OPRK1)
#Z0729N:Human Mu Opioid receptor (OPRM1)
#Z0739N: Human Cannabinoid Receptor Type 1 (CNR1)
#Z0749N: Human Delta Opioid Receptor (OPRD1)
#Z0800N: Human Angiotensin II Type I Receptor (AGTR1)
#Z0900N: Human Oxytocin Receptor (OXTR)
#Z1000N: Human C-X-C Chemokine Receptor Type 4 (CXCR4)
#Z1010N: Human C-C Chemokine Receptor Type 5 (CCR5)
#Z1100N: Human Serotonin 5HT2A Receptor (HTR2A)
#Z1200N: Human Proteinase-Activated 2 Receptor (PAR2-F2RL1)
#Z1219N: Human Proteinase-Activated 1 Receptor (PAR1-F2R)
#Z1300N: Human Lysophosphatidic Acid 1 Receptor (LPAR1)
#Z1409N: Human Parathyroid Hormone 1 Receptor (PTH1R)
#Z1419N: Human Parathyroid Hormone Receptor 2 (PTH2R)
#Z1509N: Human Serotonin 5-HT1A Receptor (HTR1A)
#Z1609N: Human Gastric Inhibitory Polypeptide Receptor (GIPR)
#Z1700N: Human Cannabinoid Receptor Type 2 (CNR2)
#Z1800N: Human Serotonin 5HT2C Receptor (HTR2C)
#Z1909N: Human Serotonin 5-HT1B Receptor (HTR1B)
#Z2009N: Human Glucagon Receptor (GCGR)
#Z2109N: Human Somatostatin Receptor Type 2 (SSTR2)
#Z2209N: Human Melanocortin 1 Receptor (MC1R)
#Z2229N: Human Melanocortin 3 Receptor (MC3R)
#Z2239N: Human Melanocortin 4 Receptor (MC4R)
#Z2309N: Apelin Receptor (APLNR)
#Z2409N: GPR17
#Z2509N: Human Atypical Chemokine Receptor 3 (ACKR3)
#Z2609N: Calcitonin Receptor-like Receptor (CRLR or CALCRL)
Gi-coupled receptors
- Human D2 Dopamine Receptor (DRD2)
- Human D3 Dopamine Receptor (DRD3)
- Human M2 Muscarinic Receptor (CHRM2)
- Human A1 Adenosine Receptor (ADORA1)
- Suman Succinate Receptor (SUCNR1)
- Human Kappa Opioid Receptor (OPRK1)
- Human Cannabinoid Receptor Type 1 (CNR1)
- Human Mu Opioid receptor (OPRM1)
- Human Nociceptin Opioid Receptor (OPRX)
- Human Delta Opioid Receptor (OPRD1)
- Human C-X-C Chemokine Receptor Type 4 (CXCR4)
- Human C-C Chemokine Receptor Type 5 (CCR5)
- Human Lysophosphatidic Acid 1 Receptor (LPAR1)
- Human Serotonin 5-HT1A Receptor (HTR1A)
- Human Cannabinoid Receptor Type 2 (CNR2)
- Human Serotonin 5-HT1B Receptor (HTR1B)
- Human Somatostatin Receptor Type 2 (SSTR2)
- Apelin Receptor (APLNR)
- GPR17
Gq-coupled receptors
- Human Calcitonin Receptor (CALCR)
- Human Oxytocin Receptor (OXTR)
- Human Angiotensin II Type I Receptor (AGTR1)
- Human M3 Muscarinic Receptor (CHRM3)
- Human Serotonin 5HT2A Receptor (HTR2A)
- Human Serotonin 5HT2C Receptor (HTR2C)
- Human Lysophosphatidic Acid 1 Receptor (LPAR1)
- Human Proteinase-Activated 2 Receptor (PAR2-F2RL1)
- Human Proteinase-Activated 1 Receptor (PAR1-F2R)
- Human M1 Muscarinic Receptor (CHRM1)
Gs-coupled receptors
- Human β2 Adrenergic Receptor (ADRB2)
- Human D1 Dopamine Receptor (DRD1)
- Human Calcitonin Receptor (CALCR)
- Human GLP-1 Receptor (GLP1R)
- Human Vasopressin Receptor (AVPR2)
- Human Parathyroid Hormone 1 Receptor (PTH1R)
- Human Parathyroid Hormone Receptor 2 (PTH2R)
- Human Gastric Inhibitory Polypeptide Receptor (GIPR)
- Human Glucagon Receptor (GCGR)
- Human Melanocortin 1 Receptor (MC1R)
- Human Melanocortin 3 Receptor (MC3R)
- Human Melanocortin 4 Receptor (MC4R)
Simple Protocol
- No cell lysis
- BSL-1 Vector
- Minimal Liquid Handling
- Low Toxicity
- Batch-transduce and freeze cells
- Benefits of Using BacMam
Don’t see your receptor of interest?
Your GPCR of interest isn’t listed? Or you have a specific modification in mind? We offer custom BacMam preparations and are more than happy to discuss your project with you. Send an email to info@montanamolecular.com.
Some common requests include:
- Alternative species and splice variants
- Constitutively expressed fluorescent proteins or genetic tags on GPCRs
- Cell-specific promoters or Cre-inducible systems to target cellular subpopulations
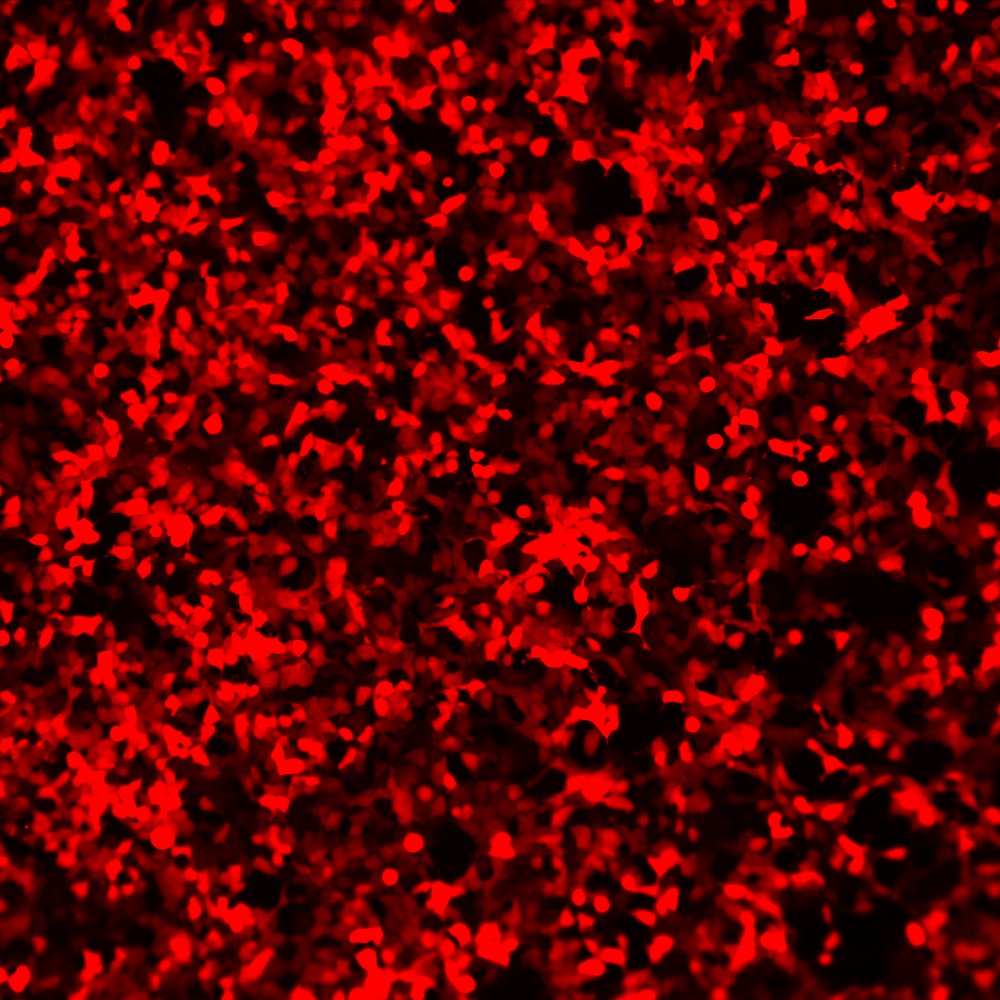
BacMam-packaged Montana Paintbrush expressing in HEK293 cells, showing even, consistent expression.
Pair with a GPCR Signaling Assay
To look at receptor signaling, our GPCR second messenger sensors for cAMP, DAG, PIP2, Calcium, and arrestin can easily be co-transduced along with your receptor.
More information on our Gs, Gi, and Gq signaling assays can be found on our GPCR Biology page, and some example data is shown below.
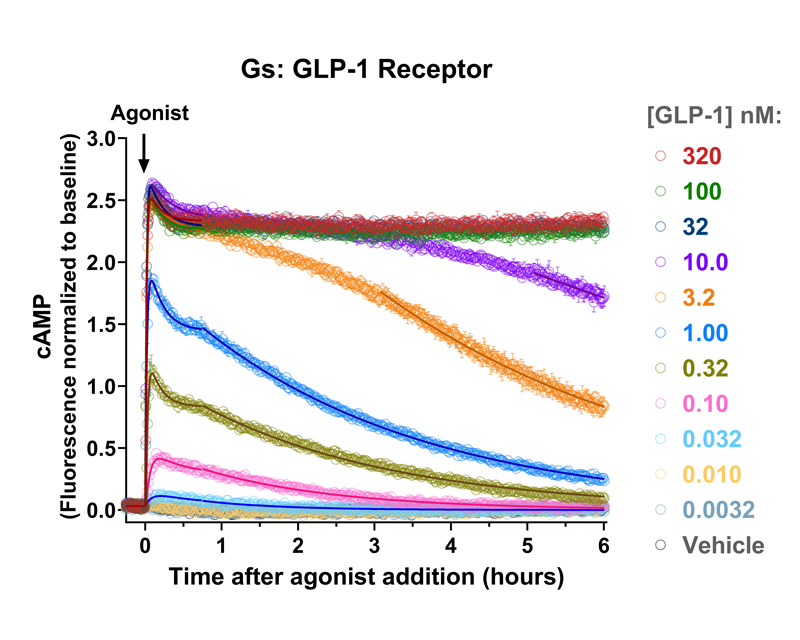
Co-expression of cAMP sensor and GLP1 receptor (#Z0600N)
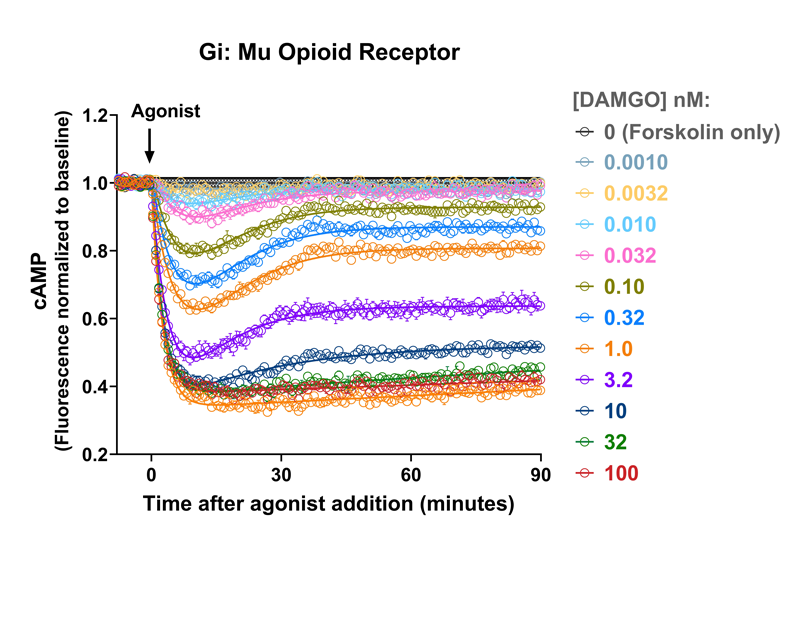
Co-expression of cAMP sensor and Mu Opioid receptor (#Z0720N)
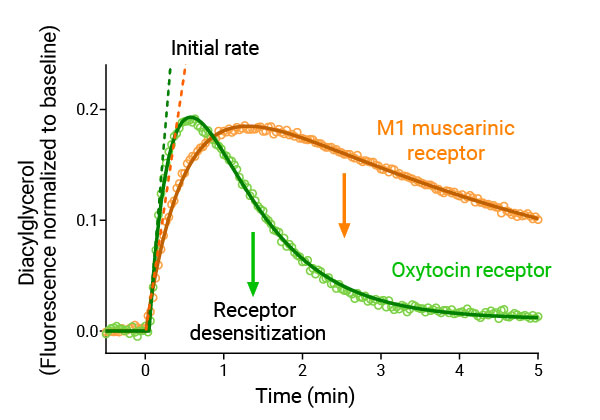
Co-expression of DAG sensor and either M1 (#Z0200N) or Oxytocin (#Z0900N) receptors
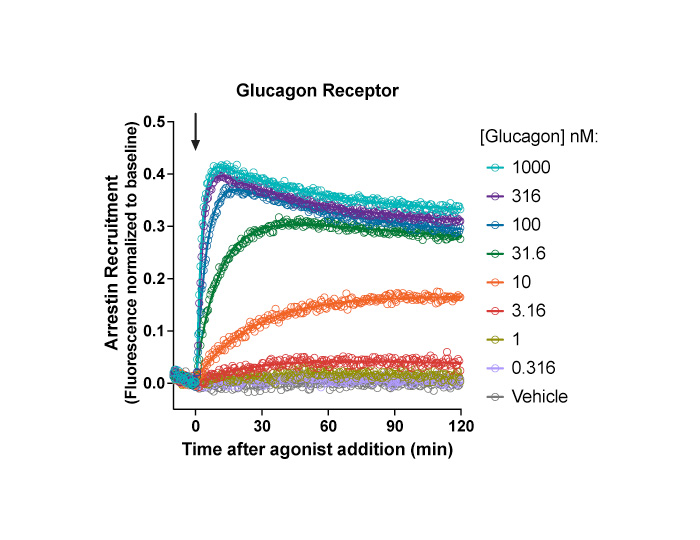
Co-expression of Arrestin sensor and Glucagon receptor (#Z2000N)
Posters
GPCR signaling kinetics in pain and OUD: A platform for measuring and analyzing the time course of signal transduction
- The timing (dynamics) of GPCR signaling impacts the physiological function and therapeutic activity of ligands activating the receptors.
- Bright fluorescent biosensors enable continuous recording of GPCR signaling in live cells, with high read frequency (seconds) for long durations (hours)
- Kinetic parameters can be extracted from the waveform data by curve fitting using a plug-in for the popular program GraphPad Prism
- This platform is applied to quantify the kinetics of partial agonism and bias at the μ-opioid receptor, and the dynamics of arrestin recruitment to multiple GPCRs.
Using the waveforms of GPCR signaling dynamics in high throughput drug discovery
- Signaling dynamics is a new frontier in drug discovery, enabling drugs to signal at the right time and in the right place.
- Signaling dynamics can be measured routinely in high throughput using biosensors, which enable continuous detection of signaling molecules over time, and the FDSS/µCell, which enables high frequency simultaneous reads of entire microtiter plates.
Measuring long term GPCR signaling and arrestin recruitment using fluorescent biosensors
GLP-1, glucagon and GIP receptor signaling dynamics were quantified using fluorescent cAMP and arrestin recruitment biosensors, identifying major differences of the duration of signaling between receptors, and between different agonists activating the GLP-1 receptor.
Publications
GPCR Assay References
GPCR Expression Vectors
- S. Shi, et al. A high-affinity, cis-on photoswitchable beta blocker to optically control β2-adrenergic receptors in vitro and in vivo. Biochemical Pharmacology. June 2024.
- A. Ippolito, et al. Increased 5-HT2A receptor signalling efficacy differentiates serotonergic psychedelics from non-psychedelics. bioRxiv. June 2024.
- L. Bridge, et al. Computational modelling of dynamic cAMP responses to GPCR agonists for exploration of GLP-1R ligand effects in pancreatic β-cells and neurons. Cellular Signaling, April 2024.
- S. Fulton, et al. Characterizing a new tool to manipulate area postrema GLP1R+ neurons across species. Physiology & Behavior. January 2024.
- M. Nuriya, et al. Alkyne-Tagged Dopamines as Versatile Analogue Probes for Dopaminergic System Analysis. Analytical Chemistry. July 2021.
- T. Vien, et al. Molecular dysregulation of ciliary polycystin-2 channels caused by variants in the TOP domain. PNAS. April 2020.
cADDis cAMP Assays
- G. Austin, et al. GLP-1R associates with VAPB and SPHKAP at ERMCSs to regulate β-cell mitochondrial remodelling and function. Nature Communications. December 2025.
- S. Chen, et al. Spatially diffuse cAMP signalling with oppositely biased GLP-1 receptor agonists in β-cells despite differences in receptor localisation. Molecular Metabolism. December 2025.
- S. Orfanos, et al. The differential effects of cAMP mobilizing agents on TGF-β-induced extracellular matrix in human lung-derived fibroblasts: insights into therapeutic targets for lung fibrosis. Respiratory Research. November 2025.
- M. Dickerson, et al. Glycolytic activation of β-cell Na+/K+-ATPases containing β1-subunits accelerates Na+ extrusion, prolonging the duration of Ca2+ oscillations but decreasing insulin secretion. Molecular Metabolism. December 2025.
- Y. Manchada, et al. Glucagon-like peptide-1 receptor C-terminal tail phosphorylation determines signalling responses from pancreatic β-cells. bioRxiv. November 2025.
- C. Metrick, et al. GPR17 structure and agonism with small molecules and oxysterols. bioRxiv. November 2025.
- S. Zervou, et al. Effect of whole-body creatine-deficiency on secretion of insulin and glucagon from isolated mouse islets. SSRN. October 2025.
- Mark Lee & Keren Hilgendorf. Prostaglandin E2 inhibits adipogenesis through the cilia-dependent activation of ROCK2. Journal of Cell Science. September 2025.
- R. Nassini, et al. Targeting prostaglandin E2 receptor 2 in Schwann cells inhibits inflammatory pain but not inflammation. Nature Communications. September 2025.
- Tatsuru Togo. Disruption of the cell membrane triggers Ca2+ mobilization using P2Y receptors and subsequent cAMP synthesis in cells surrounding wounded MDCK cells. Biochemical and Biophysical Research Communications. September 2025.
- C. Sturaro, et al. Activation of peripheral NOP receptors reduces periorbital mechanical allodynia evoked by CGRP in mice. British Journal of Pharmacology. August 2025.
- T. Cacciottolo, et al. Glucagon Receptor Deficiency Causes Early-Onset Hepatic Steatosis. Diabetes. August 2025.
- J. Chadwick, et al. Dietary-dependent sensitization of neuronal leptin signaling promotes neural repair after injury via cAMP and gene transcription. Neuron. August 2025.
- K. Berger, et al. Design of a peripherally biased NPSR1 antagonist for neuropeptide S induced inflammation. Bioorganic & Medicinal Chemistry Letters. August 2025.
- C. Gao, et al. Semaglutide drives weight loss through cAMP-dependent mechanisms in GLP1R-expressing hindbrain neurons. bioRxiv. August 2025.
- Y. Manchanda, et al. Glycaemic and bodyweight effects of GIPR coding variation reflect differences in both surface expression and intrinsic functional impairment. medRxiv. August 2025.
- S. Orfanos, et al. The differential effects of cAMP mobilizing agents on TGF-β-induced extracellular matrix in human lung-derived fibroblasts: Insights into therapeutic targets for lung fibrosis. Research Square. August 2025.
- N. Saito, et al. Activation of G protein-coupled parathyroid hormone receptors in rat incisor odontoblasts promotes mineralization via cyclic adenosine monophosphate, not Ca2+ signalling: In vitro study. International Endodontic Journal. July 2025.
- M. Merz, et al. Downstream Signaling of Muscarinic M4 Receptors Is Regulated by Receptor Density and Cellular Environment. Pharmacology Research & Perspectives. May 2025.
- B. Wysolmerski, et al. Conformational biosensors delineate endosomal G protein regulation by GPCRs. bioRxiv. May 2025.
- Paul G. DeCaen & Louise F. Kimura. Methods to Assess Neuronal Primary Cilia Electrochemical Signaling. Journal of Cellular Physiology. April 2025.
- A. Oqua, et al. Molecular mapping and functional validation of GLP-1R cholesterol binding sites in pancreatic beta cells. eLife. April 2025. (bioRxiv)
- L. Coassolo, et al. Prohormone cleavage prediction uncovers a non-incretin anti-obesity peptide. Nature. March 2025.
- R. Schuck, et al. Cholesterol inhibits assembly and oncogenic activation of the EphA2 receptor. Nature Communications Biology. March 2025. (bioRxiv)
- H. Haddad, et al. Hypothalamic opsin 3 suppresses MC4R signaling and potentiates Kir7.1 to promote food consumption. PNAS. February 2025.
- E. Blythe, et al. Endocytosis sculpts distinct cAMP signal transduction by endogenously coexpressed GPCRs. bioRxiv preprint. February 2025.
- S. Orfanos, et al. The differential effects of cAMP mobilizing agents on inhibition of TGF-β-induced extracellular matrix and growth factor expression in human lung fibroblasts. Authorea. February 2025.
- G. Austin, et al. GLP-1R associates with VAPB and SPHKAP at ERMCSs to regulate β-cell mitochondrial remodelling and function. bioRxiv. February 2025.
- A. Klein, et al. Inhibition of adenylyl cyclase 1 (AC1) and exchange protein directly activated by cAMP (EPAC) restores ATP-sensitive potassium (KATP) channel activity after chronic opioid exposure. bioRxiv. February 2025.
- I. Davies, et al. Chronic GIPR agonism results in pancreatic islet GIPR functional desensitisation. Molecular Metabolism. January 2025.
Borealis Arrestin Assays
- Y. Manchada, et al. Glucagon-like peptide-1 receptor C-terminal tail phosphorylation determines signalling responses from pancreatic β-cells. bioRxiv. November 2025.
- A. Ippolito, et al. Evidence that 5-HT2A receptor signalling efficacy and not biased agonism differentiates serotonergic psychedelic from non-psychedelic drugs. British Journal of Pharmacology. June 2025.
- E. Billard, et al. Pharmacological characterization of cannabidiol as a negative allosteric modulator of the 5-HT2A receptor. Cellular Signaling. January 2025.
- I. Davies, et al. Chronic GIPR agonism results in pancreatic islet GIPR functional desensitisation. Molecular Metabolism. January 2025.
- A. Ippolito, et al. Increased 5-HT2A receptor signalling efficacy differentiates serotonergic psychedelics from non-psychedelics. bioRxiv. June 2024.
- H. Schiff, et al. β-arrestin-biased proteinase-activated receptor-2 antagonist C781 limits allergen-induced airway hyperresponsiveness and inflammation. British Journal of Pharmacology. June 2022.
DAG Assays
- E. Lukovic, et al. Design and Evaluation of Novel Ginger 6-Shogaol-Inspired Phospholipase C Inhibitors to Enhance β-Agonist-Induced Relaxation in Human Airway Smooth Muscle. Journal of Medicinal Chemistry. June 2025.
- E. Billard, et al. Pharmacological characterization of cannabidiol as a negative allosteric modulator of the 5-HT2A receptor. Cellular Signaling. January 2025.
- X. Chen, et al. Roles for PKC signaling in chromaffin cell exocytosis. Biophysical Journal. December 2024.
- A. Hamilton, et al. Nicotinic signaling stimulates glucagon secretion in mouse and human pancreatic α-cells. Diabetes. October 2024.
- X. Chen, et al. A PACAP-activated network for secretion requires coordination of Ca2+ influx and Ca2+ mobilization. Molecular Biology of the Cell. May 2024. (bioRxiv)
- S. Datta, et al. APOL1-mediated monovalent cation transport contributes to APOL1-mediated podocytopathy in kidney disease. The Journal of Clinical Investigation. January 2024. (Supplemental Data)
- Z. Miller, et al. Lidocaine induces apoptosis in head and neck squamous cell carcinoma through activation of bitter taste receptor T2R14. Cell Reports. November 2023. (bioRxiv)
- N. Zaïmia, et al. GLP-1 and GIP receptors signal through distinct β-arrestin 2-dependent pathways to regulate pancreatic β cell function. Cell Reports. October 2023.
- G. Sanchez, et al. Coincident Regulation of PLCβ Signaling by Gq-Coupled and μ-Opioid Receptors Opposes Opioid-Mediated Antinociception. Molecular Pharmacology. December 2022.
- Z. Miller, et al. Lidocaine Induces Apoptosis in Head and Neck Squamous Cell Carcinoma Cells Through Activation of Bitter Taste Receptor T2R14. bioRxiv. November 2022.
- M. Doepner, et al. Endogenous DOPA inhibits melanoma through suppression of CHRM1 signaling. Science Advances. September 2022.
Calcium Assays
- X. Chen, et al. Roles for PKC signaling in chromaffin cell exocytosis. Biophysical Journal. December 2024.
- X. Chen, et al. In vitro and in vivo inhibition of the host TRPC4 channel attenuates Zika virus infection. EMBO Molecular Medicine. July 2024.
- X. Chen, et al. A PACAP-activated network for secretion requires coordination of Ca2+ influx and Ca2+ mobilization. Molecular Biology of the Cell. May 2024. (bioRxiv)
- C. Amos, et al. Membrane lipids couple synaptotagmin to SNARE-mediated granule fusion in insulin-secreting cells. Molecular Biology of the Cell. December 2023.
- J. Wu, et al. Interaction Between HCN and Slack Channels Regulates mPFC Pyramidal Cell Excitability and Working Memory. bioRxiv. March 2023.
- M. Thomas, et al. Optically activated, customizable, excitable cells. PLOS One. December 2020.
- L. Liu, et al. Diacylglycerol kinases regulate TRPV1 channel activity. Journal of Biological Chemistry. April 2020.
- S. Hoare, et al. A kinetic method for measuring agonist efficacy and ligand bias using high resolution biosensors and a kinetic data analysis framework. Nature Scientific Reports Feb 2020.
- K. Harlen, et al. Live-Cell Assays for Cell Stress Responses Reveal New Patterns of Cell Signaling Caused by Mutations in Rhodopsin, α-Synuclein and TDP-43 Front. Cell. Neurosci.,December 2019
- M.C. Cañizal, et al. A Dual Ca2+/ DAG Sensor Reports on Ligand Efficiency.
- C. Xingjuan, et al. PKC-dependent Phosphorylation of the H1 Histamine Receptor Modulates TRPC6 Activity. Cells. 2014.
- P. Tewson, et al. A multiplexed fluorescent assay for independent second-messenger systems: decoding GPCR activation in living cells. Journal of Biomolecular Screening 18, 2013.
- J. Wu, et al. Improved Orange and Red Ca2+ Indicators and Photophysical Considerations for Optogenetic Applications. ACS Chem Neurosci. Jun. 19, 2013
- H.J. Carlson, et al. Mutational Analysis of a Red Fluorescent Protein-Based Calcium Ion Indicator. Sensors, 2013.
- P. Tewson, et al. Simultaneous Detection of Ca2+ and Diacylglycerol Signaling in Living Cells. PLoS One. 2012.