Frequently Asked Questions
1. How do I choose the right product for my research ?
There are a few factors to consider when choosing the right product for your application. Please see our Product Pages (available from the left side menu or the footer menu) and/or contact a sales rep.
2. Why is simultaneous detection of multiple second messengers important?
Multiplex measurements in living cells tell us more about the biological response and may enable better assessments of potential adverse effects early in the drug discovery process. Cell-based calcium assays have been a mainstay of GPCR drug discovery and signal transduction research since the mid 1990s. However, a calcium readout alone is ambiguous. Many cellular processes produce calcium. Multiplex assays produce specific information about the signaling pathway and can indicate ligand bias.
3. Which of Montana Molecular's Biosensor Assays are plate reader compatible?
While fluorescence microscopy and high-content imaging systems are compatible with all of our sensors, we are proud to offer several assays that work well on automated fluorescence plate readers, with Z’ values of >0.6. These include our:
- Green and Red cADDis cAMP Assays
- Green and Red Nuclear-targeted NLS-cADDis cAMP Assays
- Green and Red DAG Assays
- Green and Red R-GECO Calcium Assays
- Green PIP2 Assay
- Green cGMP Assay
- Green and Ratiometric Cell Stress Assays
- Green Borealis Arrestin Assays
- 3CLglow and 3CLglowUp
Other assays, including those targeted to small microdomains within the cell, are best used with fluorescence microscopy and high-content imaging:
- Green and Ratiometric Cilia-targeted cADDis cAMP Assays
- Green and Red Membrane-targeted cADDis cAMP Assays
- Green ArcLight Voltage Sensor
- Red PIP2 Assay
Our customers have reported good results on the following automated fluorescence plate readers:
- Hamamatsu FDSS
- Molecular Devices FLIPR
- Molecular Devices Flexstation
- Perkin Elmer Enspire
We have validated on:
- Biotek Synergy MX
- Biotek Cytation
- BMG CLARIOstar
4. What are some experimental parameters that I can change to optimize expression in my cells?
5. What advantages do Montana Molecular's cADDis cAMP assays have over GloSensor cAMP Assay from Promega?
How does cADDis cAMP assay compare with a cAMP assay that uses luciferase?
RELEVANCE
- cADDis is available with various promoter and enhancer systems for specialized cell types.
- cADDis is packaged in a variety of viral vector systems for optimized delivery to primary cultures and iPSC-derived cells.
- cADDis does not require the addition of luciferin to produce a signal.
- Well-validated protocols for detecting Gi signaling are available for the cADDis cAMP assay.
RELIABILITY
- Luciferase is known to interact with small molecules causing about 8-10% false positive “hits” in HTS. Chem Biol. 2012 Aug 24;19(8):1060-72.
- Red cADDis can be paired with green cADDis for ratiometric imaging of cAMP signaling.
SPECIFICITY
- cADDis can be targeted sub cellularly, to compare cAMP levels in the organelle versus the cytosol.
- cADDis can be targeted to specific populations of cells via a cell-specific promoter systems including Cre-inducible expression
MULTIPLEXING
- cADDis can be multiplexed with other colored sensors, to measure two different signals, in the same cell, at the same time. Green cADDis paired with a red DAG sensors indicates when Gs/Gq bias is present. Red cADDis paired with Green ArcLight shows simultaneous cAMP and voltage signals.
6. How do I decide between an Upward DAG or cADDis assay and a Downward DAG or cADDis assay?
If you think of the signal as the absolute value of the change in fluorescence intensity, then the change in fluorescence intensity can be either increasing or decreasing regardless of whether the analyte is increasing or decreasing. We recommend the Upward sensors for imaging, because in imaging applications, the sensor is subject to more light, and the Upward signal is opposite of any potential bleaching effect. For plate reader applications, the signal is averaged over the well, so the Downward sensors are recommended because the signal from the sensor is opposite from any background fluorescence. However, both types of sensors work well in both imaging and plate reader applications. Let us know if you can’t decide as we can suggest some options for you.
7. Are Montana Molecular’s BacMam kits approved for use in my BSL-1 lab?
Yes. Our sensors are packaged in BacMam, Autographa californica, AcMNPV, which is a modified baculovirus. The virus in this kit is pseudotyped to infect mammalian cells. While the virus expresses in mammalian cells, the baculovirus genome is silent, and it cannot replicate to produce new virus in mammalian cells. While it should be handled carefully, in a sterile environment, it is classified as a Biosafety Level 1 (BSL-1) reagent.
8. Will BacMam work in my cell line?
BacMam has been validated in the following cell lines: Link to BacMam transducible cells table
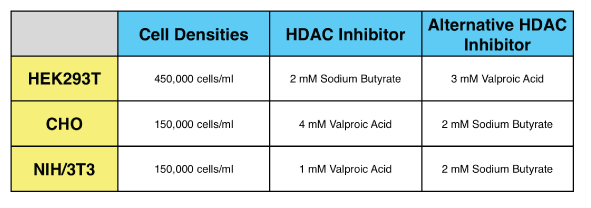
9. I am using CHO cells. Is there anything I should know in order to optimize expression in this cell type?
There are several alterations to our standard protocol that improve transduction in CHO cells. Addition of a spinoculation step is one option, replacing the sodium butyrate with Valproic Acid can help as well. A final concentration of 4-5 mM valproic acid tends to be optimal. Higher concentrations can be tested in order to further increase expression, but a decline in cell health may be observed. See our Protocols for more specifics on working with CHO cells.
Our new Big Sky BacMam also offers improved expression in CHO cells. Please contact us to inquire about using this new vector to express sensors or proteins in your cells.
10. How do I change the standard protocol for cADDis and other assays if I need to transfect a receptor?
Example: Transfecting HEK293 cells with a receptor plasmid
Day 1: Seeding cells Seed HEK 293 cells for transfection in 75cm2 flask. Aim for cells to be ≥80% confluent before transfection. OPTION 1: cells can be seeded directly in 96 well plate, if so, follow Suggestions for Assays in Adherent Cells, described in the standard protocol. Day 2: Transfect receptor:
- Ensure that cells are ≥80% confluent in flask before transfection.
- Tranfection of cells in the flask using lipofectamine2000 in OPTi-MEM.
- After 4 hrs change media. OPTION 2: transduction on Day 2, at least 1 hour after transfection.
Day 3: Transduction and Plating of cells according to the standard protocol. Assay 24-36 hours post transduction.
11. What authentication steps are taken for BacMam stocks?
Our QA/QC processes are designed to make sure your assays are reliable and results are reproducible.
BacMam stock is tested for sterility before packaging. A 100 uL sample of each BacMam stock is added to 5 mL of rich cell culture media without antibiotic and is incubated at 37°C, 5% CO2, for five days and checked for bacterial or fungal growth.
Viral genes (VG) per milliliter (mL) are measured by qPCR with primers specific to VSVG. Viral samples are prepared to release viral genomic DNA, then multiple dilutions of the preparation are run in qPCR against a standard curve to generate an average titer for each BacMam stock. Each tube of stock is labeled with VG/mL and a stock keeping unit (SKU) identifier.
When applicable, to test efficacy serial dilutions are added to cultured HEK 293 cells. After 24 hours, the number of fluorescent cells are counted to establish transducing units per mL of stock.
BacMam is stable when stored at 4°C, protected from light. However, we recommend retesting BacMam stock after storing for more than 12 months at 4°C, or after any freeze-thaw cycle.
Sterile technique should be maintained at all times when handling BacMam stocks.
12. What is the return/refund policy?
No returns will be accepted for refrigerated or frozen products, this includes assay kits and BacMam constructs. Please inspect your order and must notify us at info@montanamolecular.com within five (5) business days of delivery to report any problems as we may be able to send a replacement. Please visit the Terms of Sale page for more details.