Differential Effects of GRK2 and GRK5 on Arrestin Recruitment Kinetics
Luciana M Leo, DeLancey Doty, Sam Hoare, Anne Marie Quinn, Thom HughesPresented August 22nd at:
The G Protein-coupled Receptor Kinases and Arrestins Conference: Key Modulators of Signal Transduction
Although different GRK isoforms can enhance overall arrestin recruitment to specific GPCRs, the effect of each isoform on the kinetics of arrestin recruitment has not been systematically evaluated.
• Here, we apply a genetically encoded fluorescent biosensor and kinetic data analysis to investigate the effect of GRK2 and GRK5 on arrestin recruitment dynamics.
• For muscarinic and opioid receptors, in most cases GRK2 greatly accelerated recruitment and also induced appreciable complex degradation, whereas GRK5 produced a slower and more sustained arrestin recruitment relative to GRK2.
• Our data suggests that these receptors could be more susceptible to rapid desensitization, internalization and degradation in certain biological systems where GRK2 is highly expressed.
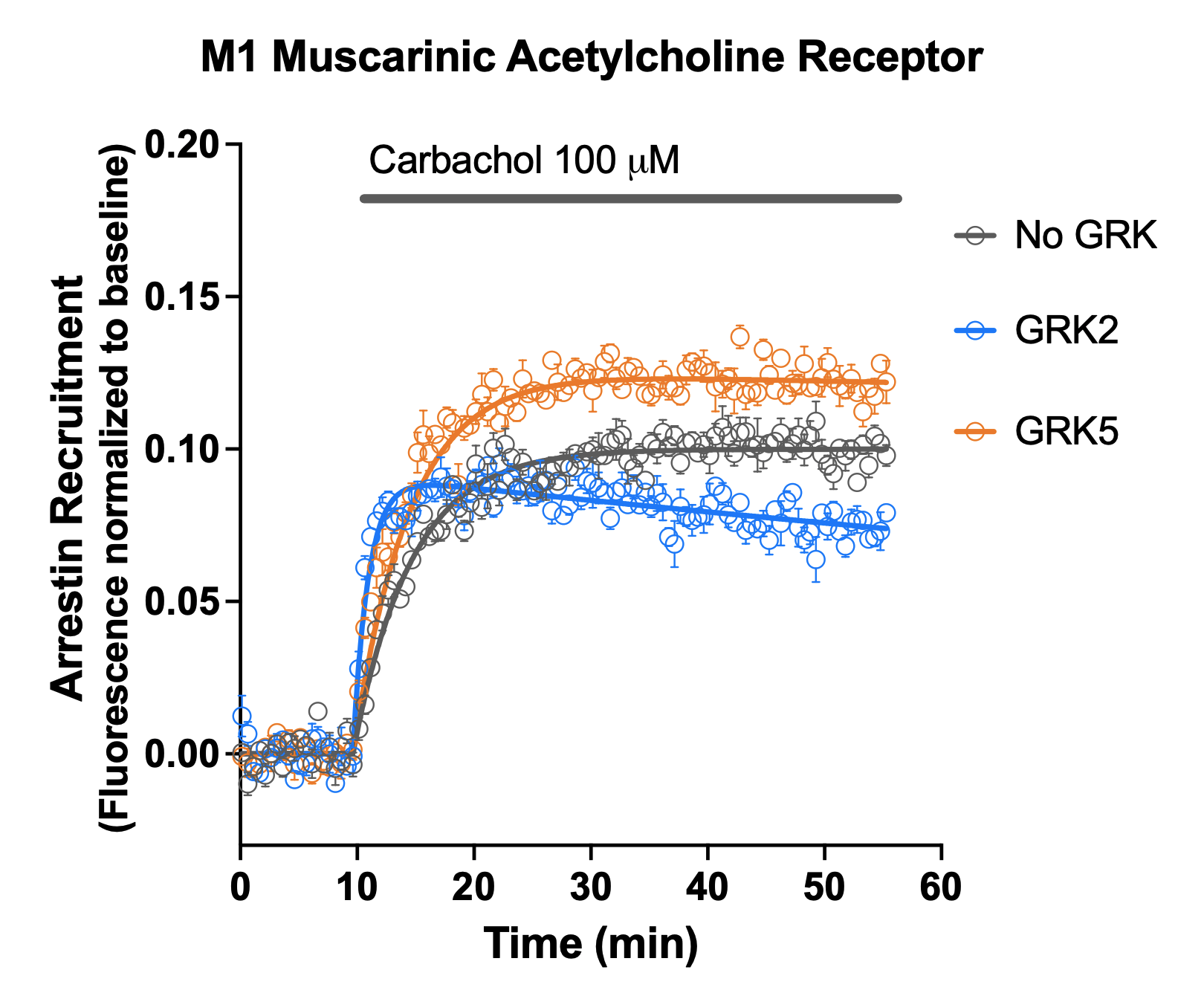
GRK Effects
M1 muscarinic receptor: GRK2 enhanced the initial rate but not the peak arrestin recruitment, and greatly increased the degradation rate (t ½ reduced from >100 to 2.5 minutes). GRK5 enhanced peak arrestin recruitment but had a smaller effect on the initial rate and degradation half-time. This indicates that GRK2 promotes fast arrestin recruitment and degradation, while GRK5 promotes a slower and more sustained arrestin signal.
Methods
- GRK isoforms or dominant negative mutants were transiently expressed in HEK293T cells along with GPCRs using BacMam vectors.
- The Borealis arrestin biosensor was used to continuously report arrestin recruitment over time.
- Curve fitting methods for kinetic data were used to extract kinetic parameters such as initial rate, degradation half-time and peak arrestin recruitment as previously described. (Hoare et al., Front. Cell Neurosci. 2022 Jan 17;15:814547).
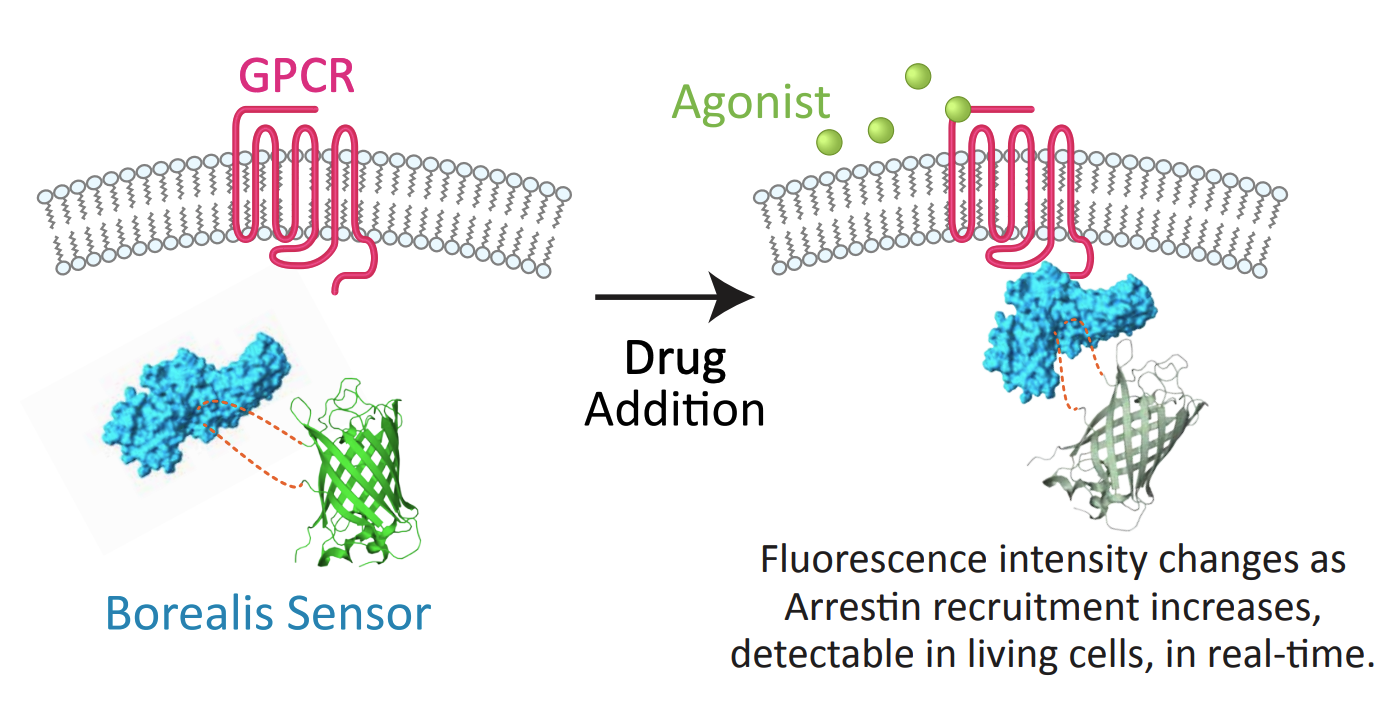
Arrestin Assay Development
Montana Molecular has recently developed a suite of fluorescent arrestin assays optimed for specific GPCRs in live cells. Please contact us if you don’t see your receptor of interest listed.



